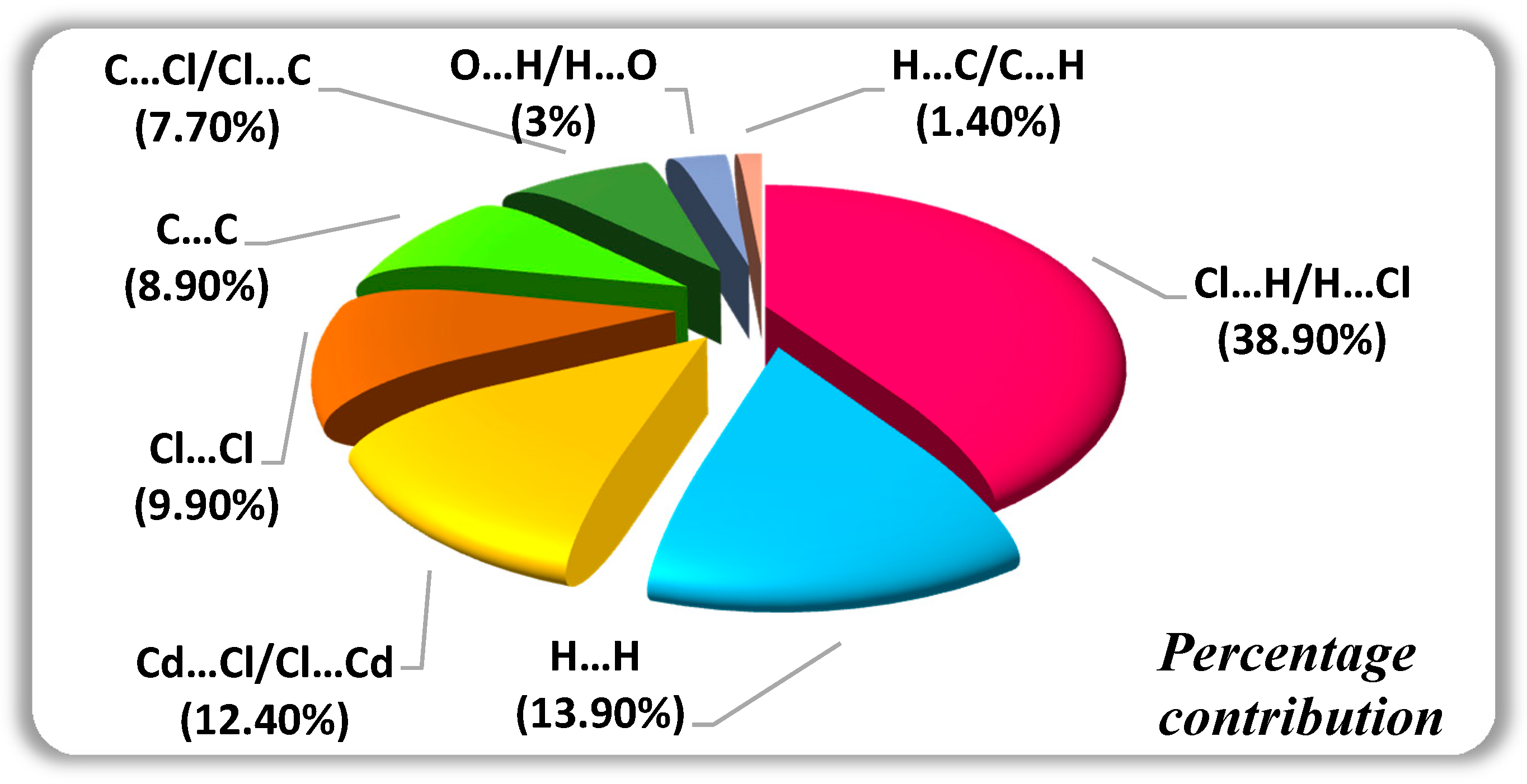
It turns out that one can obtain a lot of insight into the chemical bonding from these curves. (Curves because they are functions of energy). The study is supplemented by an application of that ML model under the scope of mechanical loading in which the resulting overview of chemical bond strengths revealed a chemical/structural heterogeneity that is in line with the tendency to bond exchange verified for atomic local environments. The above two equations define what are known, respectively, as the Crystal Orbital Overlap Population (COOP) and Crystal Orbital Hamilton Population (COHP) curves. The stated comprehensive overview is made possible through a combination of: efficient quantitative estimate of bond strengths supplied by COHP analysis, representative statistics regarding structure in terms of atomic configurations achieved with classical molecular dynamics simulations, and the smooth overlap of atomic positions (SOAP) descriptor. geometrical criteria have overlap population in the range 0.005-0.15. The connection between electronic structure and chemical bonding is given by crystal orbital Hamilton population (COHP) analysis within the framework of density functional theory (DFT).

COOP (crystal orbital overlap population) a plot analogous to population analysis for band-structure. Typically, the positive bonding region is plotted to the right of the zero line. This paper reports a machine learning (ML)-based approach that brings an unprecedented “big picture” view of chemical bond strengths in MGs of a prototypical alloy system. Overlap populations Another question is whether the filled orbitals are of a bonding or antibonding character.This is displayed on a crystal orbital overlap population (COOP) plot as shown in Figure 34.3. Here, we show that the same model and the same type of orbital-overlap arguments.
The chemistry (composition and bonding information) of metallic glasses (MGs) is at least as important as structural topology for understanding their properties and production/processing peculiarities. Single-crystal X-ray diffraction studies revealed that the complexes.
